Overview
The IMP supply chain tracks investigational product (IP) from creation through participant dispensing and return. The full workflow is available under the IMP app menu.
Navigation
| Menu | Sub-item | Purpose |
|---|---|---|
| Transfer | Receipts | Auto-generated receipts when delivery confirmed |
| Transfer | Delivery to Site | Sponsor creates delivery requests to sites |
| Transfer | Dispenses | Site records dispensing to participants |
| Transfer | Returns | Site records unused/expired IP returns |
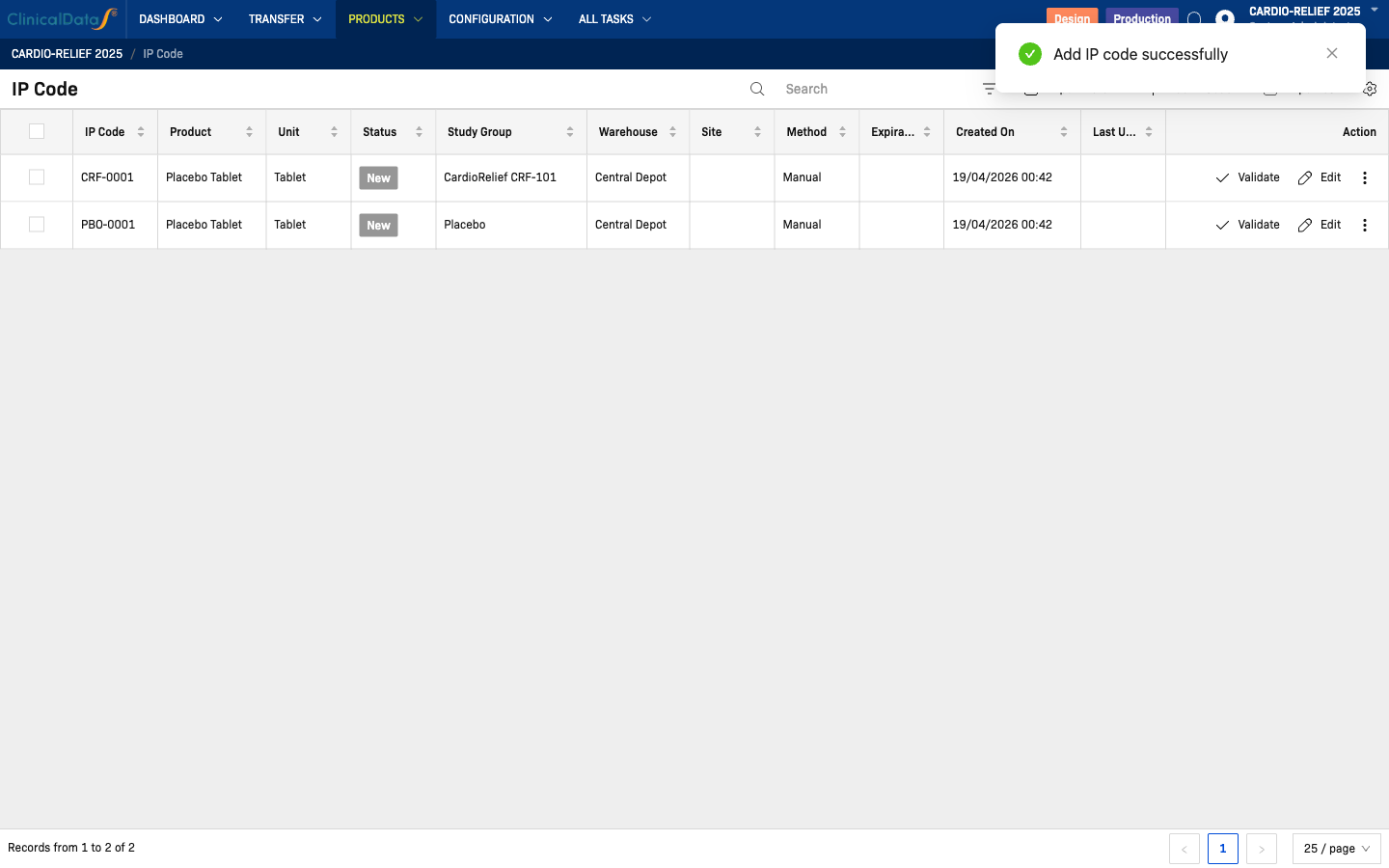
| Products | Products | View products and their barcodes |
| Products | IP Code | Add and manage individual IP codes |
| Configuration | Warehouses | Central Depot + site pharmacy locations |
| Configuration | Unit of Measure Category | Product units (e.g., Tablet) |
| All Tasks | — | Pending tasks across all transfer steps |
Supply Chain Flow
Step 1 — Configuration
└─ Warehouses: Central Depot + SITE-01, SITE-02, SITE-03 Pharmacies
└─ Unit of Measure: Tablet
Step 2 — Products
└─ Products defined: e.g., CardioRelief CRF-101 50mg Tablet, Placebo Tablet
Step 3 — IP Codes (Products → IP Code)
└─ Add codes one-by-one or bulk import via CSV
└─ Assign Study Group at creation (CardioRelief CRF-101 or Placebo)
⚠️ Unblinded operation — visible to unblinded roles only
Step 4 — Delivery to Site (Transfer → Delivery to Site)
└─ Sponsor creates Delivery Request (Central Depot → Site Pharmacy)
└─ Site confirms delivery
Step 5 — Receipts (Transfer → Receipts)
└─ Auto-generated by system on delivery confirmation
└─ Read-only list
Step 6 — Dispenses (Transfer → Dispenses)
└─ Site records IP dispensed to participant
Step 7 — Returns (Transfer → Returns)
└─ Site records unused/expired IP returned
No Country Warehouse
The ClinicalDataS IMP module uses a two-tier structure: Central Depot → Site Pharmacy directly. There is no Country Warehouse tier.
Design vs Production mode
The IMP app header shows Design and Production toggle buttons:
- Design — configuration changes allowed; no real supply transactions
- Production — live operational mode; IP codes and transfers are real
Configure all settings in Design mode before switching to Production.
Prerequisites
Before adding IP codes:
- Study randomization is configured (for Randomization purpose)
- At least one product is defined under Products → Products
- At least one warehouse is defined under Configuration → Warehouses
- Units of measure are defined under Configuration → Unit of Measure Category