Configure eConsent from Home → select your study → Pre-Screening and eConsent → e-Consent Settings tab.
Configuration requires the study to be in Design status. Settings are read-only for Available or Frozen studies.
Enable e-Consent Features
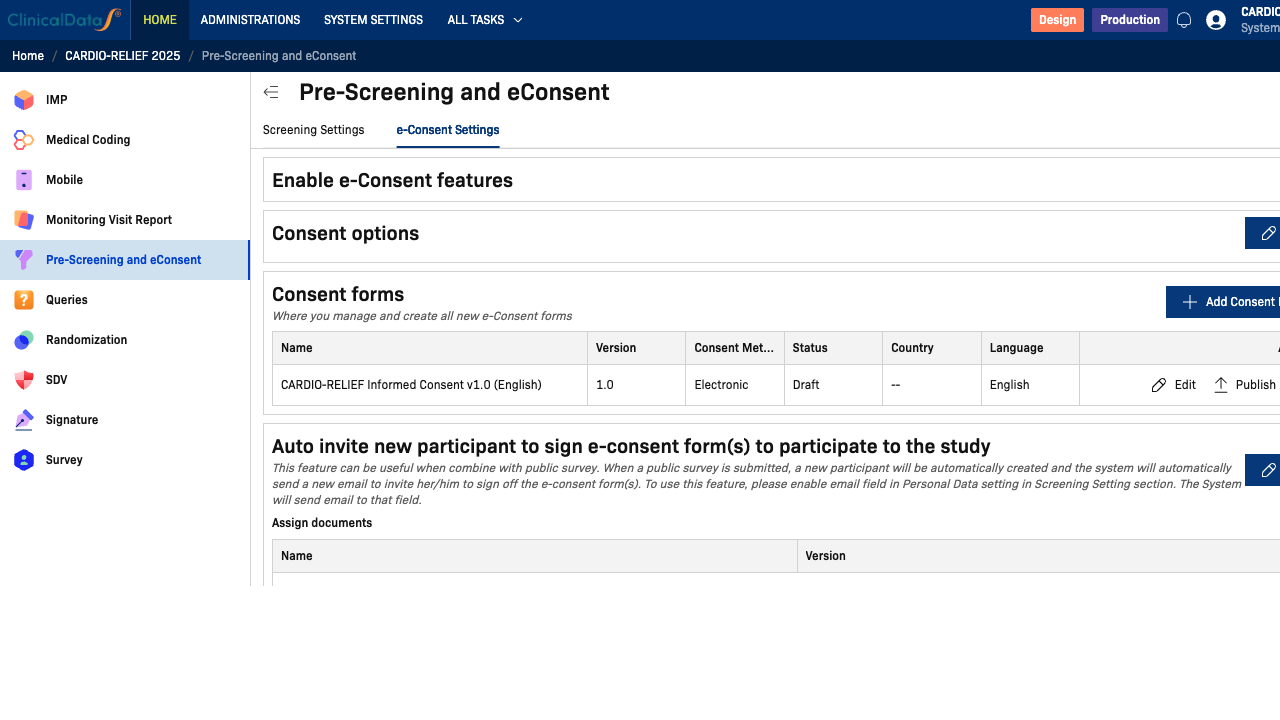
Toggle Enable e-Consent features to activate the electronic consent workflow. When enabled, three additional sections appear:
- Consent options — general consent behavior and notification settings
- Consent forms — manage and create ICF documents
- Auto invite — automatically send consent invitations to new participants
Consent Options
Click Edit in the Consent options section to configure behavior such as consent expiry rules and reminder notifications. Save your changes to apply.
Consent Forms
The Consent forms table lists all consent form versions for the study. Each row shows:
| Column | Description |
|---|---|
| Name | Consent form title |
| Version | Form version number |
| Consent Method | Electronic or Paper |
| Status | Draft, Published, or Archived |
| Country | Country scope (if set) |
| Language | Language of the consent form |
| Action | Edit, Publish, or additional options |
Add a Consent Form
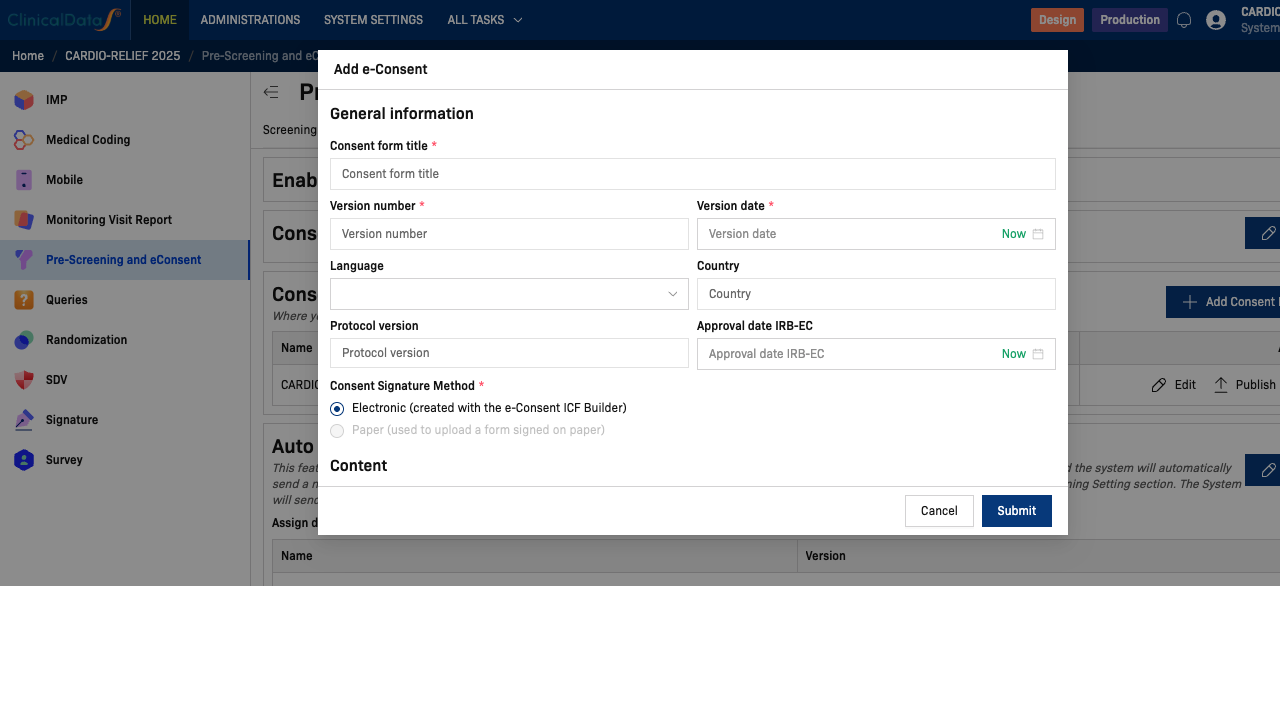
Click + Add Consent Form to open the Add e-Consent dialog. Fill in the following fields:
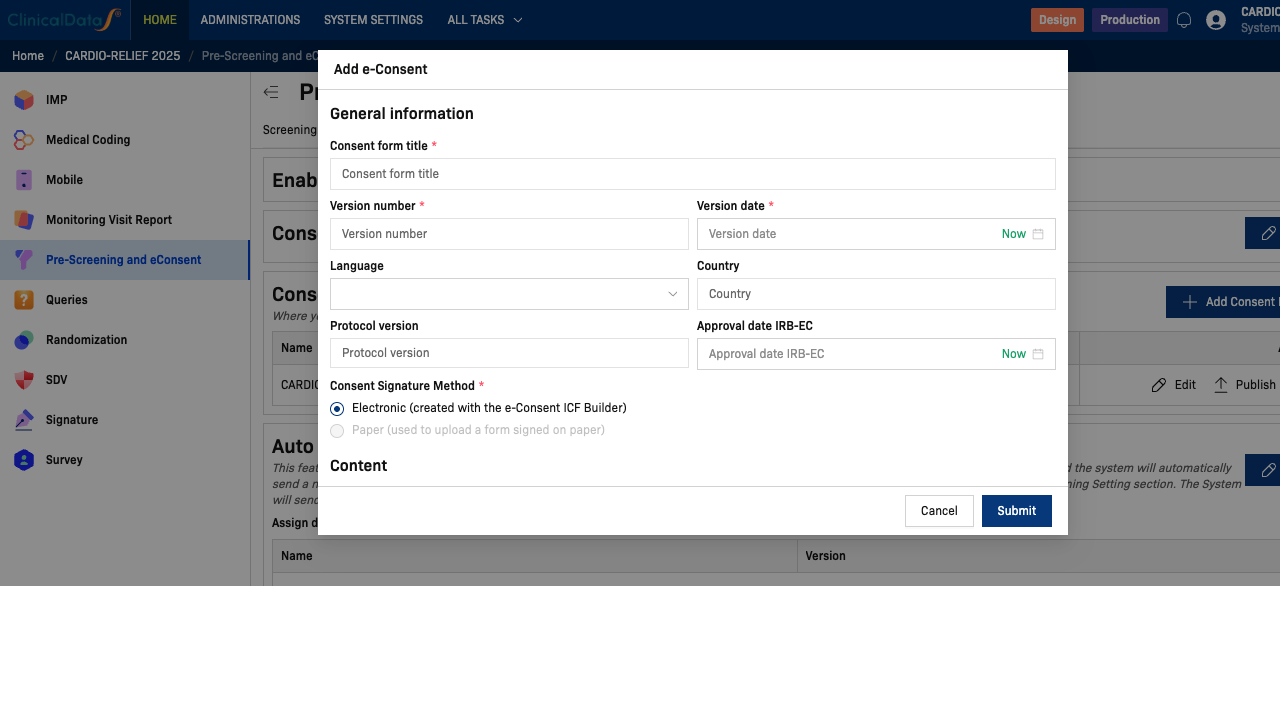
General Information
| Field | Required | Description |
|---|---|---|
| Consent form title | ✅ Yes | Full title of the ICF (e.g., CARDIO-RELIEF Informed Consent v1.0 (English)) |
| Version number | ✅ Yes | Version of the consent document (e.g., 1.0) |
| Version date | ✅ Yes | Date of this version. Click Now to auto-populate today's date |
| Language | No | Language of this form (e.g., English, Vietnamese) |
| Country | No | Country scope for this form |
| Protocol version | No | Associated protocol version (e.g., CDS-2025-CR-001 v1.0) |
| Approval date IRB-EC | No | IRB or Ethics Committee approval date. Click Now to auto-populate |
| Consent Signature Method | ✅ Yes | Electronic (built with the ICF Builder) or Paper (uploaded scan) |
Content
| Field | Required | Description |
|---|---|---|
| Content of Consent | ✅ Yes | Full ICF body text. Uses a rich text editor supporting headings, bold, lists, tables, and links. Add all study sections (purpose, procedures, risks, benefits, confidentiality, contacts, etc.) |
Setting — Participant Questions
Click Add Question to add checkbox questions participants must review before signing.
Setting — Participant Agreement
Click Add Participant Agreement to add statements participants must acknowledge before signing.
Setting — Participant Signature
| Field | Required | Description |
|---|---|---|
| Subject Signature Label | ✅ Yes | Label shown for the participant's signature (e.g., Participant Signature) |
| Signature Status | ✅ Yes | Require (default) or Optional |
| Subject Signature statement | ✅ Yes | Confirmation statement the participant attests to when signing (rich text) |
Setting — Legally Authorized Representative (LAR)
Click Add LAR Question to add LAR-specific questions when a legal representative signs on behalf of the participant.
Setting — Study Team Member Section
Click Add Team Member to add investigator or study staff signature sections (maximum 2 team member signatures).
Click Submit to save the consent form as a Draft. A draft form can be reviewed and edited before publishing.
Publishing a Consent Form
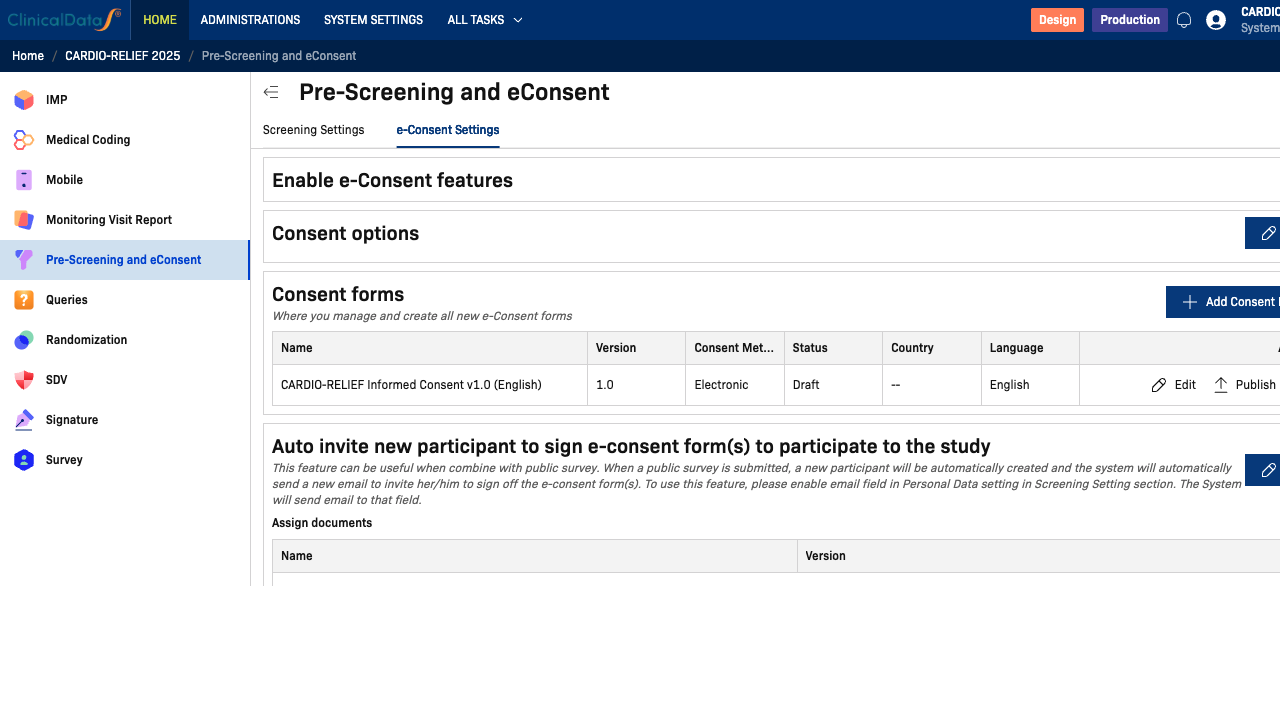
From the Consent forms table, click Publish in the Action column to activate the consent form. Published forms are available for participant enrollment. Use the ⋮ menu to archive or duplicate a form.
Auto Invite
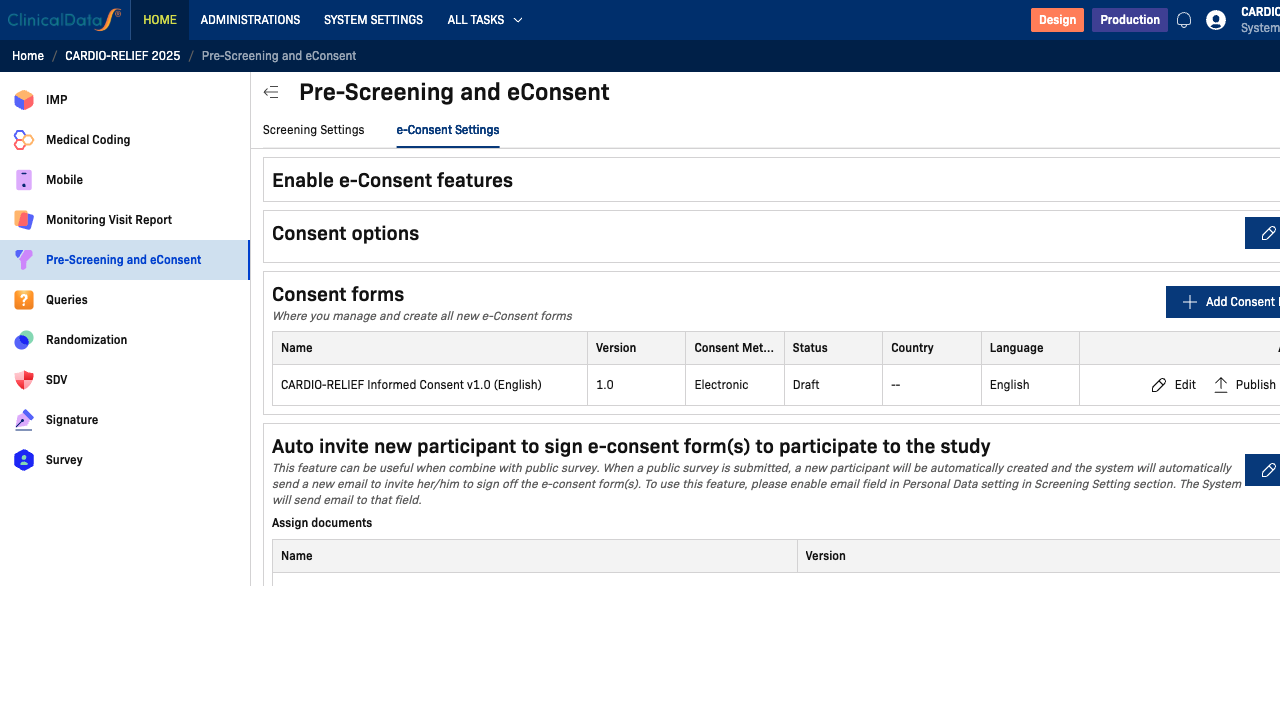
The Auto invite section automatically sends a consent invitation email to new participants when they are created via a public survey submission.
To use Auto invite, the Email field must be enabled under Collect Personal Data in the Screening Settings tab. The invitation is sent to the email address collected during screening.
Click Edit in the Auto invite section to assign which consent form documents should be sent automatically. The Assign documents table shows which consent forms are linked to the auto-invite workflow.